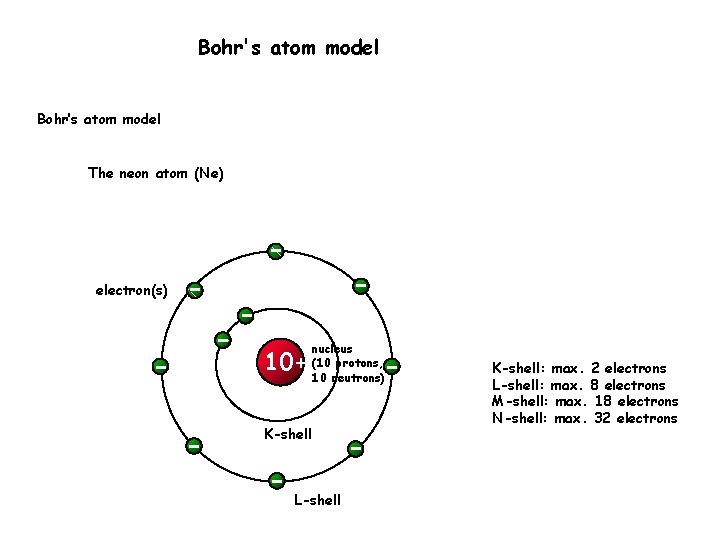
The singular proton of the hydrogen atom or the protons and neutrons of the hydrogen-like species(ex- He +, Li 2+, etc.) are present in the centre of the atom, in a positively charged nucleus.Hydrogen Bohr model was based on some assumptions which are listed below: Deriving Energy of an Electron in a Stationary Stateīohr’s Atomic Model was for the hydrogen atom and hydrogen-like species(species with only one electron).Deriving Velocity of an Electron in a Stationary State.As a result, Rutherford’s Atomic Model doesn’t work. Atoms don’t form and simultaneously blow up. The electron spirals moving closer to the nucleus eventually would collapse into it in what calculations suggest a time of 10 -8 s.īut life still lives on. Thus, negatively charged electrons would emit energy as they revolve around the nucleus, with its orbit decreasing with time. Maxwell’s Laws of Electrodynamics suggest that a charged particle moving in a circular orbit releases energy in the form of electromagnetic radiation. It was like our solar system, with planets revolving around the sun, a beautiful symmetry that sadly wasn’t feasible. The Rutherford Atomic Model placed the protons and neutrons inside a small structure called the ‘nucleus’ with the electrons revolving around the nucleus in circular orbits. Discovery of the proton, electron, and neutron, made us wonder, what does the atom actually look like? With his classic, Alpha Ray Scattering Experiment Rutherford was able to suggest a possible model for the atom. Research, experiments, and theory into the atomic structure have resulted in much understanding of the stuff that makes us.ĭalton’s Atomic Theory, which stated that atoms are indivisible was shattered with the discovery of subatomic particles, which make up the atom. 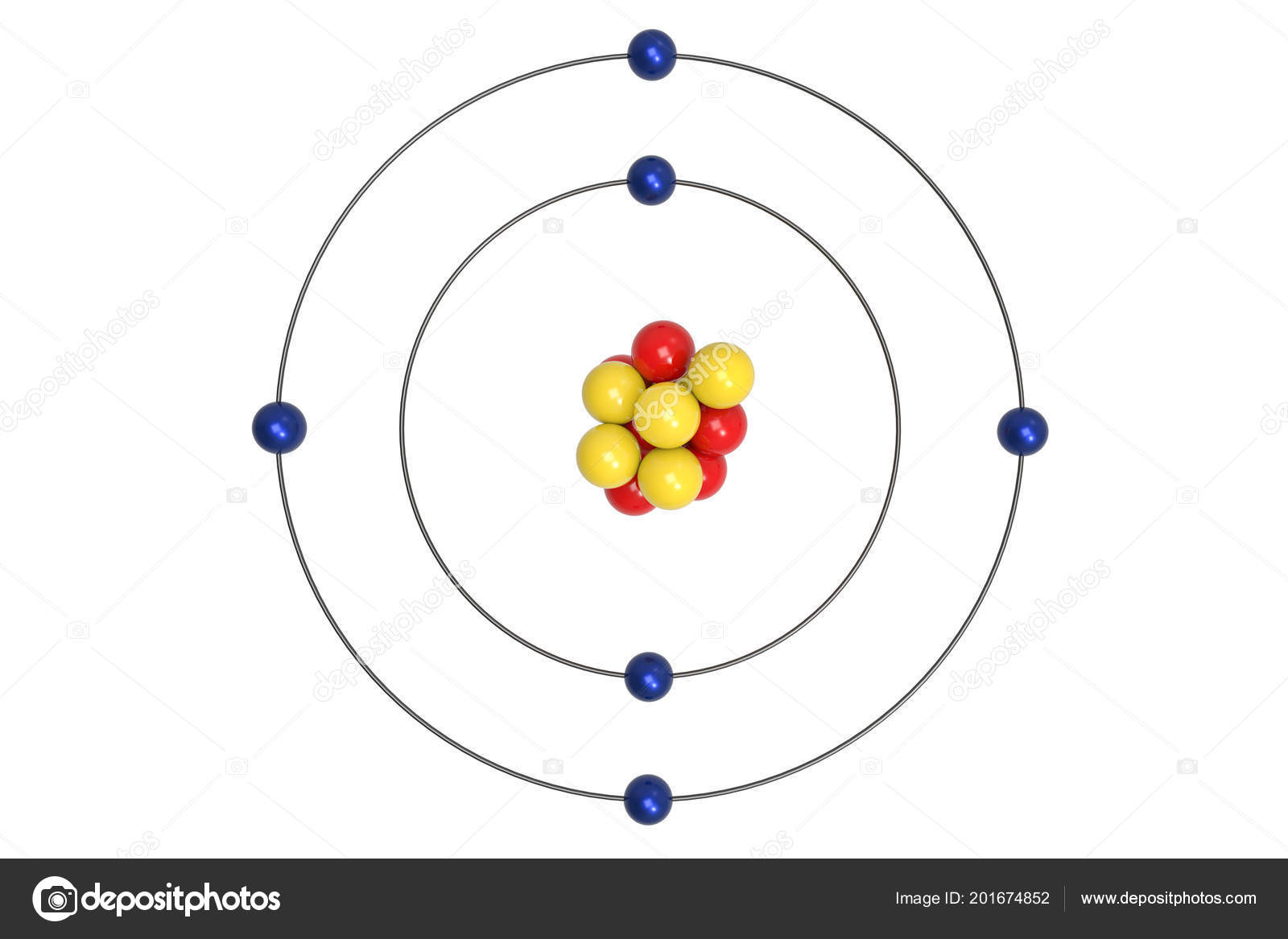
Let us look at a bit of history and then get into the details of this model. It was basically a modified version of Rutherford’s Atomic Model wherein Bohr explained that electrons move in fixed orbitals (shells) and not anywhere in between and he also explained that each orbit (shell) has a fixed energy level. Bohr’s Atomic Model was introduced by Niels Bohr in 1915.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
